What The FDA Doesn't Want You To Know: Cookie Recall LEAKED!
Have you ever wondered what really happens behind the scenes when food products get recalled? The truth might surprise you. When major cookie brands suddenly disappear from shelves without much public explanation, it raises serious questions about food safety transparency. Today, we're diving deep into a shocking cookie recall that the FDA would rather keep quiet, revealing what manufacturers don't tell consumers and why these dangerous omissions keep happening.
The Hidden Dangers in Your Cookie Jar
A popular cookie brand has abruptly yanked six different kinds of treats off shelves after failing to disclose potentially deadly ingredients. This isn't just about a simple labeling mistake – it's about consumer safety being compromised for profit. The Food and Drug Administration (FDA), which is supposed to protect American consumers, recently issued a critical product recall for several Gina Marie Bakery cookie products distributed from their Waterbury, Connecticut facility.
What makes this situation particularly alarming is how these recalls often fly under the radar. Most consumers never hear about them until it's too late, or they find out only after experiencing allergic reactions. The FDA's communication strategy around food recalls has been criticized for being too passive, leaving vulnerable populations at risk.
- Asia Argentos Epstein Connection The Nude Photos And Cover Up They Tried To Bury
- 180 Minutes To Hours The Leaked Secret Thats Gone Viral
The Amazon Fresh Cookie Recall: A Class I Emergency
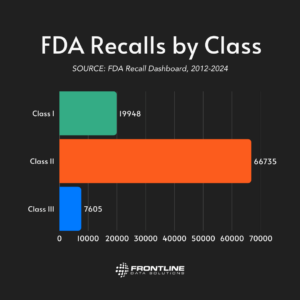
The Amazon Fresh cookie recall is an FDA Class I recall due to undeclared egg allergens. Class I recalls represent the most serious type of recall, where there is a reasonable probability that the use of or exposure to a violative product will cause serious adverse health consequences or death. This classification alone should have triggered widespread public notification, yet many consumers remained unaware.
Here's why it took time to surface and if it may still pose a risk. The lag between discovery and public notification often spans weeks, during which time contaminated or mislabeled products continue circulating in the marketplace. For individuals with severe allergies, this window of uncertainty can be life-threatening. The FDA's current recall notification system relies heavily on voluntary compliance from manufacturers and retailers, creating gaps in consumer protection.
Connecticut Company's Major Packaging Failure
A Connecticut company has recalled various cookie products because of possible undeclared allergens, the Food and Drug Administration (FDA) announced. This recall wasn't initiated due to contamination or spoilage – it was a complete failure in the packaging and labeling process that put thousands of consumers at risk.
- You Wont Believe What Happened After I Tried This Viral Malibu Bay Breeze Recipe
- Epsteins Dark Secret Never Before Seen Nude Corpse Photos Surface
The recall was announced after the company discovered a major issue with its packaging. It failed to list some important ingredients that could trigger severe allergic reactions. This isn't a minor oversight; it's a fundamental breakdown in quality control that suggests deeper systemic problems within the manufacturing process.
Specifically, the cookies may contain almonds, sesame, and several food colorings without declaring them on the label. Each of these ingredients represents a significant allergen risk. Almonds are among the most common tree nut allergens, sesame has been recognized as a major allergen requiring specific labeling, and certain food colorings have been linked to behavioral issues and allergic reactions in sensitive individuals.
The FDA's Communication Crisis
The FDA is here to provide the facts, and you can help by sharing this information with your community and loved ones. However, the agency's current approach to public notification has significant limitations. Their recall database exists, but it's not easily accessible to the average consumer, and updates aren't systematically pushed to the public through mainstream channels.
Did you know some drugs that aren't approved by the FDA are still on the market? These unapproved FDA drugs were grandfathered onto the market ages ago, creating a parallel system where consumers can't easily distinguish between rigorously tested products and those that have never undergone modern safety evaluations. This grandfathering loophole extends beyond medications into various consumer products, undermining the FDA's core mission of protecting public health.
Lead Contamination and Other Hidden Hazards
To determine if the level of lead in a food is a potential health concern, the FDA considers the toxicity of lead and potential exposure based on the level of lead measured in the food and other factors. Lead contamination in food products represents another area where FDA oversight has been criticized as insufficient. The cumulative effects of low-level lead exposure, particularly in children, can cause developmental delays, learning difficulties, and behavioral problems.
The agency's threshold for action often allows levels of contaminants that many health experts consider unsafe. This "acceptable risk" approach means that products containing harmful substances may remain on shelves as long as they fall below certain limits, even when those limits are higher than what independent research suggests is truly safe.
Beauty Products and Carcinogen Concerns
Acne treatments from brands including Proactiv and Clinique have elevated levels of the carcinogen, an independent testing lab said in a petition filed with the FDA. This revelation highlights how FDA regulation extends beyond food into cosmetics and personal care products, yet the agency's authority in these areas is surprisingly limited. Unlike drugs and medical devices, cosmetics don't require pre-market approval, and the FDA's ability to mandate recalls is restricted.
An overview of FDA's authority over cosmetics under the Federal Food, Drug, and Cosmetic Act and the Fair Packaging and Labeling Act reveals significant gaps in consumer protection. The agency can take action only after problems arise, rather than preventing dangerous products from reaching consumers in the first place.
The Quaker Oats Contamination Crisis
The Quaker Oats Company today announced an expansion of the December 15, 2023, recall to include additional cereals, bars and snacks listed below because they have the potential to be contaminated. This recall, which expanded multiple times as the scope of contamination became clear, demonstrates how initial recall notices often underestimate the problem's true scale.
The Quaker Oats Company today announced the recall of specific granola bars and granola cereals listed below because they have the potential to be contaminated with salmonella, an organism which can cause serious and sometimes fatal infections in young children, elderly people, and others with weakened immune systems. Salmonella contamination represents one of the most common and dangerous food safety issues, yet the response time from both companies and regulators often leaves consumers exposed for extended periods.
Medical Device Recalls and Regulatory Challenges
Info related to recall of medical devices, corrections and removals shows that FDA oversight extends far beyond food products. Medical device recalls can be particularly complex, as they often involve implanted devices that require surgical removal or replacement. The agency's Medical Device Correction and Removal program aims to address these issues, but the process can be slow and bureaucratic.
Regulations and federal register notices govern how recalls must be conducted and communicated, but these regulations often prioritize corporate interests over consumer protection. The legal framework allows companies significant discretion in how they handle recalls, including decisions about timing and scope of notifications.
The Phenylephrine Phase-Out Controversy
The Food and Drug Administration recently announced plans to phase out phenylephrine, the key ingredient in some cold and allergy pills. This decision came after decades of the ingredient being widely available, raising questions about how long potentially ineffective or harmful substances remain on the market before regulatory action is taken.
The phase-out process itself highlights another challenge: even when the FDA identifies problems, removing harmful products from the market can take years. During this transition period, consumers continue using products that regulators have deemed ineffective or potentially dangerous.
USDA and FDA Coordination Challenges
Department of Agriculture (USDA) and U.S. Food and Drug Administration (FDA) are listed in the widget. The division of food safety oversight between these two agencies creates a fragmented regulatory landscape. Some foods fall under FDA jurisdiction while others are regulated by USDA, leading to potential gaps in coverage and inconsistent safety standards.
To find a specific recall, you can scroll through the items listed in the widget and click on the recall or alert for more information. However, this process assumes consumers know where to look and have the time to search through databases. The lack of proactive notification means that many recalls go unnoticed by the people most at risk.
The Politico Investigation: FDA Under Fire
A Politico investigation based on more than 50 interviews finds the FDA is failing to meet American consumers' expectations on food safety and nutrition. This comprehensive investigation revealed systemic problems within the agency, including understaffing, outdated technology, and a culture that often prioritizes industry interests over public health.
The investigation highlighted how the FDA's reactive approach to food safety means that problems are typically addressed only after people get sick or die. This "wait and see" strategy contrasts sharply with more proactive approaches used in other countries, where preventive measures and rigorous pre-market testing are standard practice.
Conclusion: Taking Control of Your Food Safety
The cookie recall that sparked this investigation is just one example of a much larger problem with food safety oversight in America. While the FDA plays an important role in protecting consumers, the agency's limitations mean that individuals must take additional steps to protect themselves and their families.
Start by learning to recognize recall notifications and checking products before consumption, especially if you or family members have allergies. Consider supporting organizations that advocate for stronger food safety regulations and more transparent recall processes. Most importantly, share information about recalls and safety concerns with your community – because when it comes to food safety, knowledge truly is power.
The next time you hear about a product recall, remember that it likely represents just the tip of the iceberg. Behind every announced recall are countless other products that may have similar issues but haven't yet been identified or disclosed. By staying informed and demanding better from both manufacturers and regulators, consumers can help create a safer food system for everyone.

:format(webp)/cdn.vox-cdn.com/uploads/chorus_asset/file/15412478/force-590-15.0.1435149867.jpg)